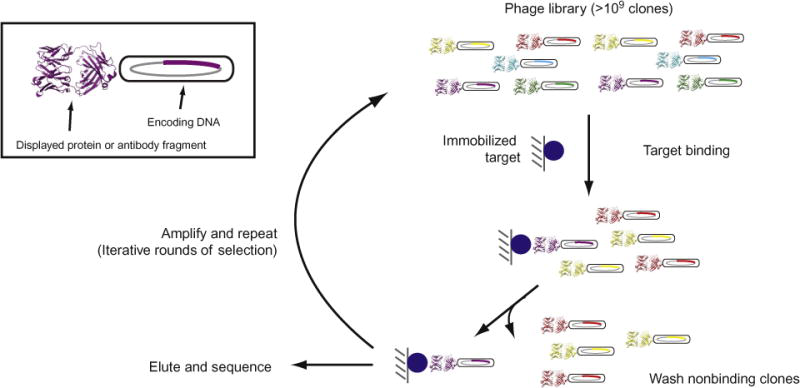
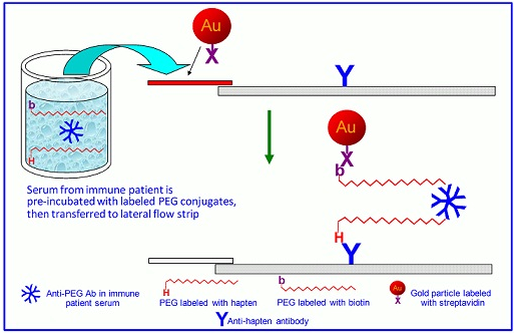
lateral flow assay development
Experience with lateral flow assays andor liquid assays in drugs of abuse highly desired. Thats why we offer end-to-end lateral flow cassettes manufacturing customized to.
50 Nitrocellulose An assortment of flow rates already cut into ready to use strips 30 Sample Pads.

. Youve come to the right place. Ad 80 of errors occur at point of sampling. Lateral Flow Assay Development Guide Lateral flow assays LFAs are rapid and inexpensive diagnostic devices that can be used to test for a target substance analyte in a sample.
40 Backing Cards. Know your target molecule. They consist of a sample pad.
Lateral Flow Assay Design. Manufacture Documentation Pilot Batch. Taking a lateral flow test strip from the design stage through product development to final manufacturing is a process that employs principles from biology chemistry physics and engineering.
Standardizing membrane characteristics and optimizing molecular level immunoassay reaction between analyte and detector molecules. Design Requirements typically describe the function. The low-stress way to find your next lateral flow assay development job opportunity is on SimplyHired.
Hands-on experience developing assays to semi-quantitative or qualitative drug levels anti-drug antibodies and pharmacodynamic measures of activity using immunoassays andor other methodologies. The lateral flow assay LFA is one of the most popular technologies on the point-of-care diagnostics market due to its low cost and ease of use with applications ranging from pregnancy to environmental toxins to infectious disease. While the use of these tests is relatively straightforward significant development time and effort.
Our services span all phases of the development cycle from feasibility to manufacturing. The SMV coat protein CP was. In the preliminary phase the reaction specificity of capture and detector anti.
30 Cellulose Absorbent Pads. We understand your urgency and need for high-performance lateral flow assembly. NanoComposix is a full-service lateral flow development company specializing in the design and manufacture of ultra-sensitive quantitative diagnostic assays.
See salaries compare reviews easily apply and get hired. Low development costs and ease of production of LFAs have resulted in the expansion of its applications to multiple fields in which. Universal Lateral Flow Assay Kit ab270537 is designed to enable simple and rapid development of proof-of-concept sandwich lateral flow immunoassays.
Eliminate errors with improved design. Core Assay Development Starter Kit includes. Whether you require the complete outsourced service or just one aspect of our service we offer a partnership approach which allows you to work with one.
Assays are designed based on these requirements which are referred to as Design Requirements or Design Inputs. Top Lateral flow assay with serial dilutions of hormone analyte showing the increase in sensitivity when using gold nanoshells blue as the probe over 40 nm spherical gold red. Standardizing membrane characteristics and optimizing molecular.
DCN Dx develops lateral flow flow-through dry chemistry and other assays for rapid diagnostic tests around a variety of conjugate labels and reporting requirements. Facing such difficulties the de-novo development of an aptamer based LFA for small molecule detection remains a challenge. The kit includes everything needed for lateral flow assay development with any pair of capture and detection antibodies.
Designed for complete in-the-field use. Our lateral flow development expertise is paired with our market-leading gold nanoparticle technology or detector label of your choice to deliver an assay to meet your market requirements. What are lateral flow assays.
The lateral flow assay LFA is a paper-based platform for the detection and quantification of analytes in complex mixtures where the sample is placed on a test device and the results are displayed within 530 min. The Future of Lateral Flow Assembly. Feasibility Report Feasibility prototype tests.
Lateral flow immunoassay systems are generally single-step assays requiring only the addition of a sample. There are over 56 lateral flow assay development careers waiting for you to apply. Our Lateral Flow Assembly services are engineered to handle your lateral flow test strip orders from the design phase to final manufacturing.
Experience interpreting and reporting complex. Soybean mosaic virus SMV is the most common virus in soybean and poses a serious threat to crop production and germplasm recession in many countries worldwide. It combines universal LFA strips Lightning-Link and.
In this best practices blog we highlight ten important considerations when developing your LFA which we hope will give you a deeper insight into lateral flow assays to streamline your development process. It is an antibiotic substance belonging to the penam class of beta lactam antibiotics. Rapid Diagnostic Labels and Reporting for Assay Development.
4444 4 5 LATERAL FLOW COMPONENTS Figure 2. The development of Lateral Flow Immunochromatography Assay can be divided into two levels. 56 lateral flow assay development jobs available.
Lateral Flow Assay using different. Development assay characterisation Final manufacturing documentation erification and validation activities final VV report final QC specification. A test design that works well in the RD lab is of little use if it is difficult to manufacture reliably in high yield.
The same structure is used whether the product development is being performed on behalf of a customer contract assay development or it an in-house product developmentWithin this structure there is a degree of flexibility and. Investigation and development of assay format particle conjugation membrane coating strip architecture. Lateral Flow Development and Manufacturing Journey Quality Partnership Results Innovation Shaun Phillips RD Senior Scientist Contract RD overview.
Optimisation and characterisation of materials formulations and processes. We have chosen ampicillin as a small molecule model for developing an aptamer based LFA. Before embarking on the development of a lateral flow assay the critical requirements of the assay you are going to produce should be outlined.
30 Glass Fiber. He holds a BS in human biology and a BA in clinical psychology from the University of California San Diego. The development of Lateral Flow Immunochromatography Assay can be divided into two levels.
The biology of your target molecule will be the key factor in developing the biology of your test. Some of the advantages to the LFA format are that these assays have a long shelf life do not require refrigerated storage and a result can be obtained in a short. Thoughtful lateral flow assay design and development is key to high performance and minimized costs achieved in part by selecting the best components for optimal assay results.
RD from concept to commercialization. He spent several years characterizing surface markers on cancer stem cells for potential use as. In this study a highly practical and rapid lateral-flow assay LFA was developed for the detection of SMV.
The Core Assay Development Starter Kit comes with all that you need to begin optimizing your assay development. Lateral flow assays are an inexpensive solution for preventative medicine. Lateral flow rapid test development at Abingdon Health typically follows a number of stages with formal reviews at critical points.
Verification and validation of manufacture process. Andre Alfaro is the director of assay development at nanoComposix.

Rotavirus Antigen Rapid Test Rotavirus Acute Gastroenteritis Gastroenteritis

Healthcare Edi Market Size Trends And Forecast To 2026 Health Care Patient History Marketing Set

Lateral Flow Assay Reader Gm Design Development Health Care Design Development Medical

Dcn Diagnostics Lateral Flow Assay Development Youtube Opportunity Analysis Marketing Trends Marketing

Rapid Test Kits Creative Diagnostics

Lateral Flow Assay Based Point Of Care Poc Testing Is Gaining Prominence Market Research Report On Lateral Flow Assa Point Of Care Testing Infographic Care

آلة تصنيع الكبة السورية Youtube تصنيع

Urinary Catheters Market Size Trends And Forecast To 2027 Catheter Urinary Incontinence Incontinence

Cory Sulieman On Twitter Cardiovascular Disease Cardiovascular Disease

Dcn Diagnostics Lateral Flow Assay Development Youtube Opportunity Analysis Marketing Trends Marketing